By Amaka Yusuf and Dara Ajala-Damisa (Lead writers)
The World Health Organisation has reported that each year, between 5.7 and 8.4 million people die due to poor quality care in low- and middle-income countries (LMICs) and this accounts for 15 percent of overall deaths. Majority of affected LMICs record lost productivity due to poor quality care, indicating that about $1.4 to $1.6 trillion is lost annually.
Why Quality Maternal Health Medicines is a Critical Issue
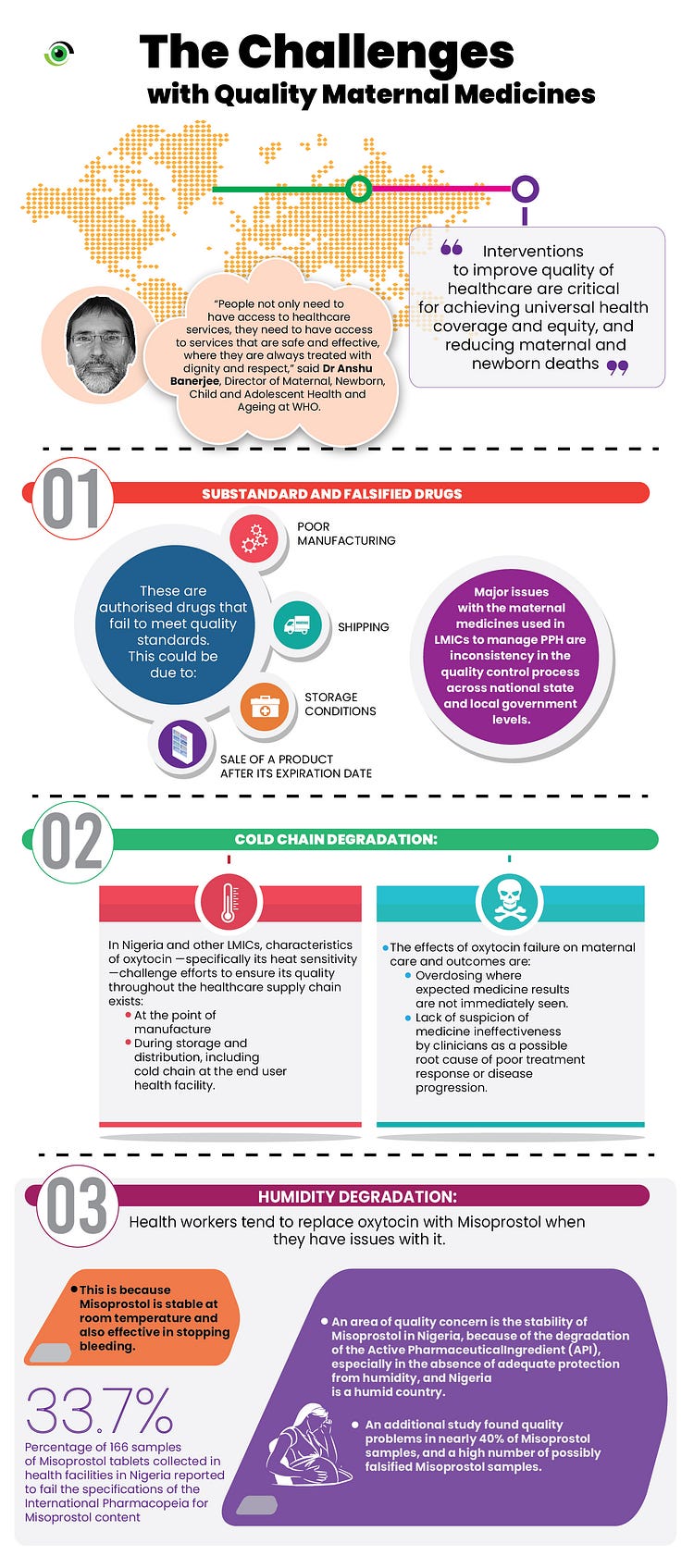
Quality care is a critical issue for maternal and new-born health. It is estimated that 6 in 10 neonatal conditions and half of maternal deaths in poorer countries are due to poor quality services. As of 2018, based on the NDHS Survey, it is estimated that there are 512 maternal deaths per 100,000 live births. The causes of maternal mortality can be categorised as either medical, socio-economic or cultural. It is estimated that 70 percent of maternal deaths in Nigeria are as a result of one of these complications: bleeding after child birth, sepsis (life threatening complications from an infection), unsafe abortion, hypertensive diseases associated with pregnancy, and obstructed labour. Generally, there are concerns about the quality of medicines available in LMICs for treating excessive bleeding, eclampsia and sepsis (life threatening complications from an infection).
This is critical to note since clearly, the quality of maternal health medicines has an important impact on quality of care which pregnant women receive. This quality of care is also directly linked to Nigeria’s maternal health outcomes which sadly still rank as one of the worst in the world.
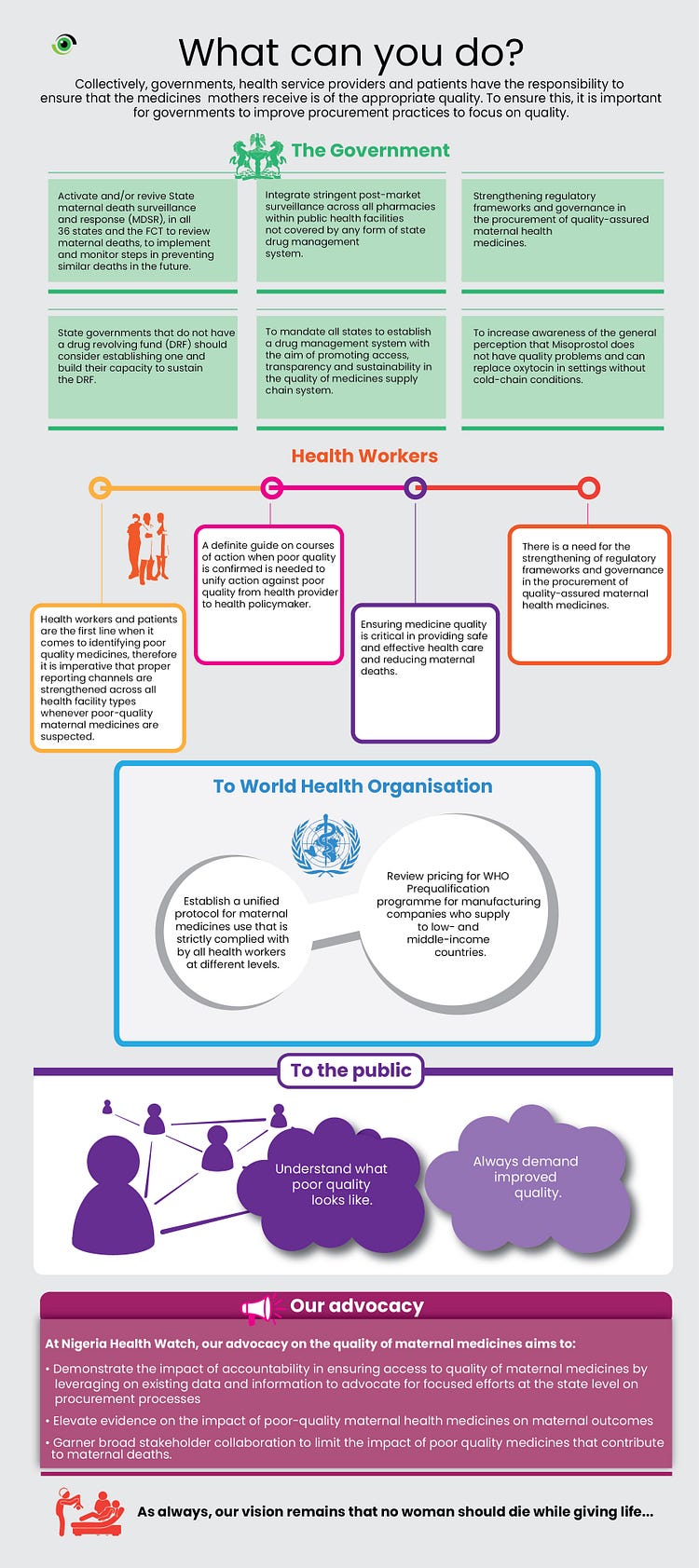
The goal of this article is to inform advocacy to promote efficiency in the procurement, management, and distribution process at all levels of government from national, state to local to ensure maintenance of standards in the delivery of medicines to the communities.
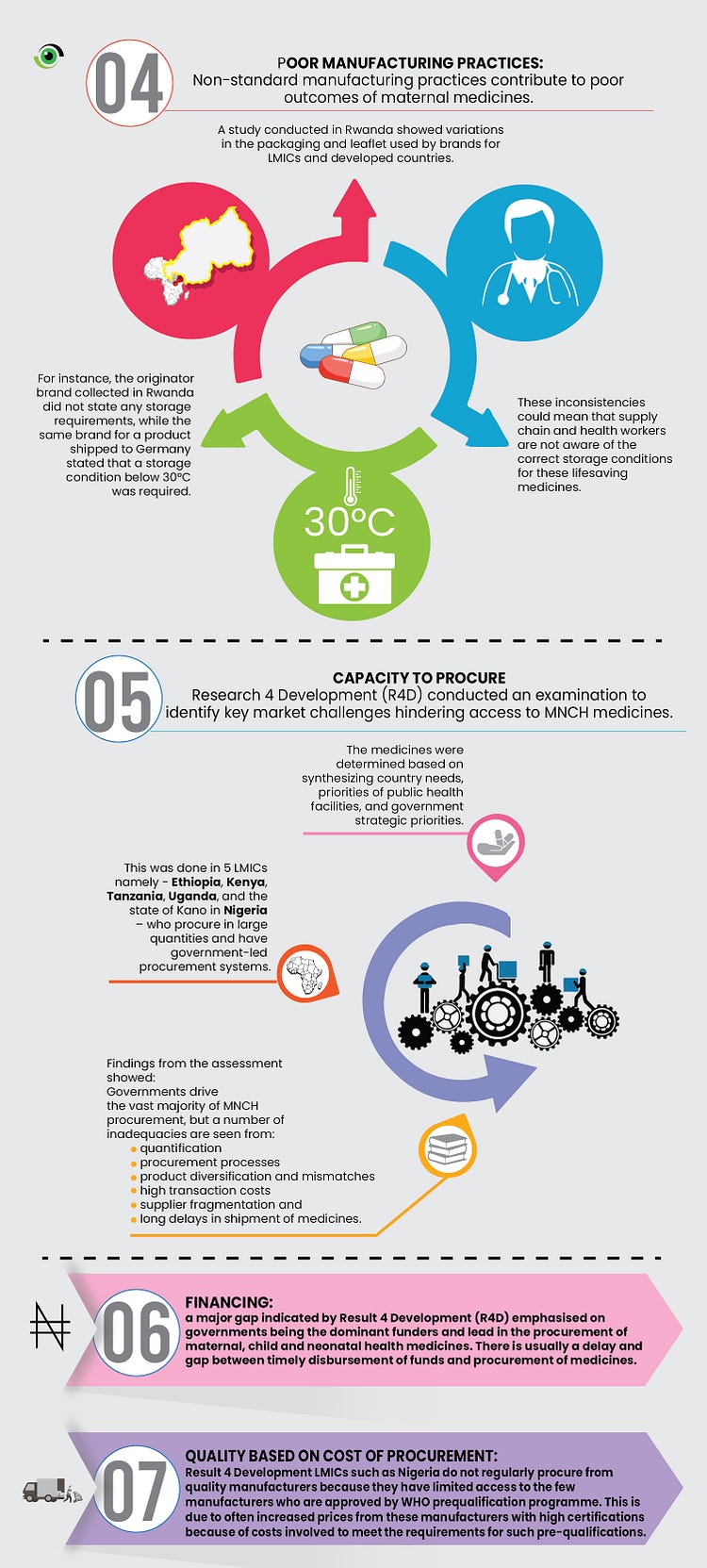
Fortunately, deaths from excessive bleeding, eclampsia and sepsis can be avoided yet these are attributed to the major cause of maternal deaths. Following a rigorous literature review to understand the reasons behind these deaths, there were clear indications that there could be variations that exist in the quality of medicines produced for LMICs and the pharmacovigilance system in the country.