Digital technology has become an integral part of daily life, and healthcare delivery can be improved by taking full advantage of the interconnectedness that technology affords.
To examine ways digital health is leveraged to improve access to medicines in Nigeria, Salient Advisory in partnership with Nigeria Health Watch organised a webinar titled, ‘Innovations in Digitising Health Products Distribution: Exploring the landscape and opportunities in Nigeria.’
The Covid-19 pandemic highlighted the important role of innovation in health systems, said Vivianne Ihekweazu, Managing Director, Nigeria Health Watch, in her welcome remarks. This, she added, underlined the critical need to digitise and bring efficiency to the pharmaceutical industry, as well as improve supply-chain and last-mile distribution.

Discussions at the webinar explored Salient Advisory’s 2022 report titled, ‘Innovations in Digitizing Distribution of Health Products: Current Trends In Nigeria, Ghana, Kenya & Uganda’ This market intelligence report is part of a longitudinal study carried out in Kenya, Ghana, Nigeria, and Uganda.
An overview of some key findings and recommendations of the report are presented in this article.
Growth Trends
- More than 80 companies are digitising the distribution of health products in Nigeria, Uganda, Kenya, and Ghana. These innovators offer a range of services such as, digitising distribution to providers and consumers, and product data services. The trend showed that innovators who digitise distribution to providers appear to be growing, with a strong user retention rate estimated at 46%.

- In 2022, in addition to start-ups and e-commerce giants, physical pharmacies also launched online operations.
- Digitising distribution to the consumers remains of high interest to innovators, but the impact of distribution on over- the- counter and prescription products Is not yet clear.
- Innovators offering product data services are few, but well established; with counterfeit drug detection and track and trace services being the most common offerings.
- All four focus countries have dedicated efforts to strengthening online pharmaceutical regulations over the past 12 months. These regulations are developing in real time and will have an impact on business models and ecosystem growth.
Funding Trends
- Recent investment momentum has been strong across the ecosystems in the focus countries, as 36% of all external funding ever raised entered the ecosystem in the last 12 months.
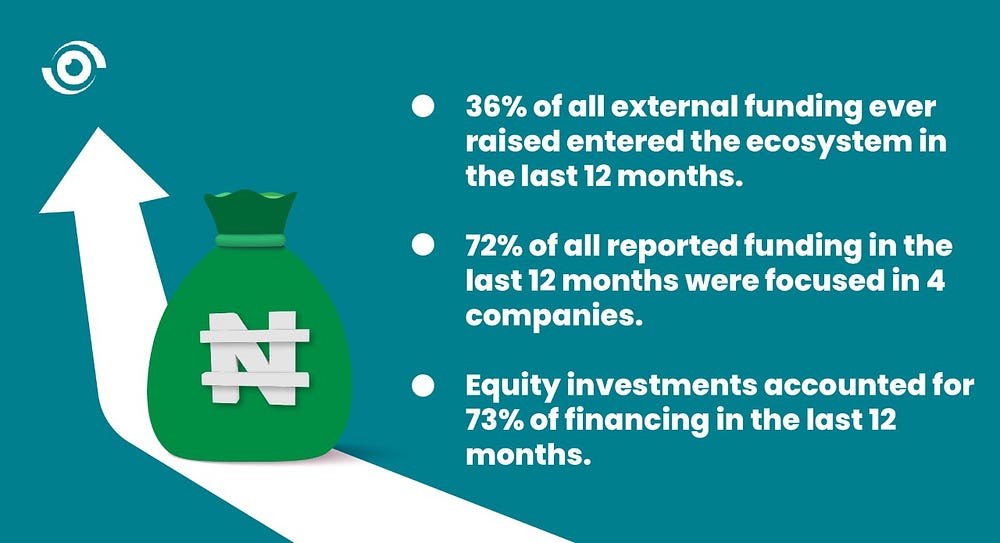
- Even though funding is growing, it is highly concentrated. 72% of all reported funding in the last 12 months were focused in 4 companies. Such a high concentration of funding in a few high-profile companies could lead to gaps in the ecosystem. This is something that needs to be closely monitored and avoided in the future.
- Equity investments accounted for 73% of financing in the last 12 months. This shows that investors are confident in the promise and commercial viability of African health start-ups.
- Exclusionary trends have remained evident over the past year, with the highest impact on race and gender. At the intersection of race and gender, black women founders have only been able to raise $1.6 million (only 2%) of funding in the last 12 months.
- Local funding sources are emerging, and 58% of the innovators that raised funding in the last 12 months cite Africa led investors as a source of funding. Over the last 12 months, there has been an over 50% increase in the number of active Africa led investors in the space. This trend may be leveraged, as donors seek to channel funds optimally to investors.

Recommendations
The African tech and digital health ecosystem are maturing, which provides the opportunity for focused action to support innovators who are well-positioned to scale their operations and impact. This can be done in the following ways:
- Partnerships with innovators demonstrating transformative potential is key. The time is now to connect the top innovators to customers in donor-funded agencies, governments, and industry. This will help them establish pilots to distribute subsidised medicines and other critical health products, explore their potential to reduce pricing, ensure affordability, and improve visibility.
- Major purchasers like governments, donors, and donor funded agencies should articulate critical distribution needs and create mechanisms to purchase innovations that fit their specifications.
- Establish a multi-country regulatory working group and provide demand-driven technical assistance in support of countries’ fast-moving efforts to define and implement regulations for digitised distribution and related services such as telemedicine.
- Support leading governments to test the aggregation of global, regional, and local supply chain data to develop pharmaceutical data platforms, including data from digitised distributors operating in the private sector, to inform public health supply chain planning and broader visibility.
- Test if and how early-stage digital payment innovations can facilitate the engagement of pharmacies in the delivery of primary care.
While regulation, which will help guide the digital pharmaceutical ecosystem and its growth trajectories, is still taking shape, efforts are still required to make sure that it remains innovation friendly, both in the short and long term.
It is hoped that this report will galvanise global health actors to prioritise focused action to support innovators who are well positioned, to help scale operations and further drive impact.


